 The Group has pioneered the physics of semiconducting polymers as light emitting diodes. The Group is deeply involved in understanding the basic physics of the light-emitting processes which involves techniques such as excited state spectroscopy.
The Group has pioneered the physics of semiconducting polymers as light emitting diodes. The Group is deeply involved in understanding the basic physics of the light-emitting processes which involves techniques such as excited state spectroscopy.
The research program is built on a broad scientific approach aiming to understand the electronic properties of polymer semiconductors in general, but also aiming to understand in-depth the physical processes that govern the performance of polymer electronic devices. A particular important issue is related to the electronic structure of interfaces between two different polymers, such as in polymer blends used in many state-of-the art polymer light-emitting or photovoltaic diodes, or a semiconducting and a gate dielectric polymer at the active interface of a field-effect transistor.
 In a polymer LED light is emitted when positive hole and negative electron charge carriers are injected into the organic semiconductor metallic contacts made to opposite sides of the semiconducting polymer films. When electrons and hole come under the influence of their mutual Coulomb attraction inside the device they recombine with each other emitting a photon. The wavelength of the emitted light depends on the band gap of the semiconducting polymer.
In a polymer LED light is emitted when positive hole and negative electron charge carriers are injected into the organic semiconductor metallic contacts made to opposite sides of the semiconducting polymer films. When electrons and hole come under the influence of their mutual Coulomb attraction inside the device they recombine with each other emitting a photon. The wavelength of the emitted light depends on the band gap of the semiconducting polymer.
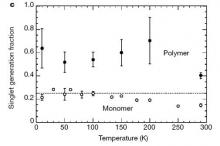
Electron and holes are spin-1/2 particles, and the exciton that is formed can either be a 'singlet' exciton or a 'triplet' exciton. From simple spin statistics one would expect 25% of singlet excitons and 75% triplet excitons to be formed if the spin of the incoming charge carriers is random. In a polymer material with weak spin-orbit coupling only the singlet excitons can recombine radiatively, since the ground state is a singlet state. From this one would expect that the efficiency of polymer LEDs cannot exceed 25%. However, in practice many polymer LEDs exceed this limit, and we have recently been able to show that the process of electron-hole capture leading to exciton formation is spin-dependent. Approaches to improve the performance of polymer LEDs include the incorporation of heavy atoms to increase spin-orbit coupling and enable harvesting of triplet excitons.
As an alternative to indium tin oxide, to reduce contact resistance and to improve lifetime of devices, we are investigating the use of metal oxides (notably zinc and molybdenum oxide) deposited by a variety of methods.
Performance of green LEDs can exceed 50 cd/A, with an external quantum efficiency of 15%.
