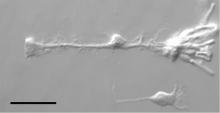
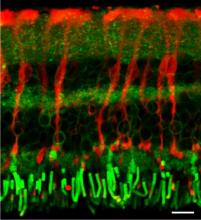
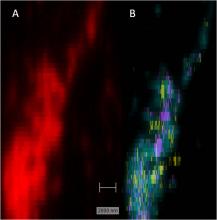
A strength of the research in BSS is the combination of expertise in optical microscopy, image analysis and techniques such as spectroscopy or micromanipulation. This is one of the core experimental areas where the potential for new collaborations is great.
We have a wide range of upright and inverted microscopes available. Almost all of them are equipped with either phase contrast or differential interference contrast (DIC). This allows the direct observation of living samples, which otherwise would be characterized by poor contrast. Most of our microscopes are also suited for epifluorescence imaging.
We also possess a state-of-the-art confocal laser scanning microscope (cLSM). This microscope offers the unique opportunity of highly sensitive fluorescence measurements with an excellent spatial resolution. A resonance scanner allows imaging with up to 100 frames per second. The system also has a built in white-light laser source for excitation. In combination with acousto-optic filters this enables us to use almost any dye currently available. Future developments based on this system include laser scanning absorption microscopy (LASCAM) and coherent anti-Stokes Raman scattering (CARS) microscopy.
A key area of development is label-free imaging techniques for biological applications. We have recently built a CARS microscope which can perform fast label-free imaging as well as broadband spectroscopy. Additionally it can perform multiphoton fluorescence microscopy. In this area we are working on additional non-linear techniques such stimulated Raman spectroscopy (SRS) for bio-imaging applications and are collaborating with life scientists across the university.
Nanospectroscopy and imaging using surface-enhanced techniques is an area of considerable expertise and has immense possibilities for developing chemical & bio-sensors for applications in healthcare and diagnostics. Surface-enhanced Raman spectroscopy (SERS) besides being employed for ultra-sensitive detection applications is also being developed for chemical mapping of cells.
Image analysis of video data from optical microscopy is an area of strength and there is a growing work in this field. The group is working on:
- quantifying the mechanics and dynamics within egg cells
- tracking the emergence of forces during the dorsal closure of drosophila
- measuring critical properties and dynamics in cell membranes
- understanding and quantifying ciliary motion in the respiratory tract
- particle tracking to study local rheological properties (see microrheology)
This area requires advanced optical microscopes, fast video acquisition, high definition and experience in image analysis algorithms.