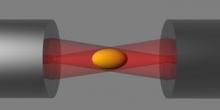
Optical Tweezers are instruments built on the principle that a tightly focused laser beam can be used to hold an object. Very often the object is a micron sized particle with a higher refractive index than the surrounding liquid. In a typical experiment, the holding force on the particle is known. The bead is then brought into contact with the object to be studied, and the forces induced on the bead can be measured. This technique has been very fruitful in a wide spectrum of problems, including understanding molecular motors, interaction potentials in colloid systems, and the mechanics of biological cells. The tweezer systems in BSS is very advanced: tens of beads can be manipulated simultaneously, with experiments fully automated through the video feedback. They are being used to study mechanics of red blood cells, phagocytosis, hydrodynamical colloidal interactions, and nonequilibrium statistical physics.
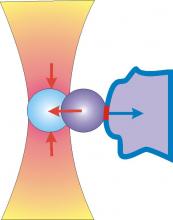
The optical stretcher is a novel laser tool that can be used to trap and deform individual biological cells. The forces arise from the momentum transfer of light to the surface. The deformation can be used to study the cytoskeleton of cells, which is an internal polymer gel resisting the external forces. Changes in the cytoskeleton are diagnostic for pathological changes and can be detected with the optical stretcher, which includes being able to distinguish between normal and cancer cells. The group is aiming to develop a label-free, high-throughput cell analysis method for cancer diagnosis and stem cell sorting.
Optical Tweezers are instruments built on the principle that a tightly focused laser beam can be used to hold an object. Very often the object is a micron sized particle with a higher refractive index than the surrounding liquid. In a typical experiment, the holding force on the particle is known. The bead is then brought into contact with the object to be studied, and the forces induced on the bead can be measured. This technique has been very fruitful in a wide spectrum of problems, including understanding molecular motors, interaction potentials in colloid systems, and the mechanics of biological cells. The tweezer systems in BSS is very advanced: tens of beads can be manipulated simultaneously, with experiments fully automated through the video feedback. They are being used to study mechanics of red blood cells, phagocytosis, hydrodynamical colloidal interactions, and nonequilibrium statistical physics.
Particle tracking microrheology. Video imaging to follow sub-micron sized beads in complex fluids, and the analysis of their fluctuation spectrum and correlations, permits study of local rheology and its heterogeneity. This is being used to study gelation in protein solutions and clay dispersions, and the local anisotropy of viscoelastic linear response in aligned gels of protein filaments. Additionally we investigate the inside of living cells as they undergo a variety of processes.